Discovered and patented1 at the University of Toronto by EBT’s co-founder Professor Paul Yoo, saphenous neuromodulation presents totally new possibilities for treating overactive bladder.
Saphenous neuromodulation (SAFN) works via a centralized mechanism, making it ideal for intermittent stimulation rather than continuous. This novel target offers numerous advantages over existing neuromodulation targets, including the tibial nerve.
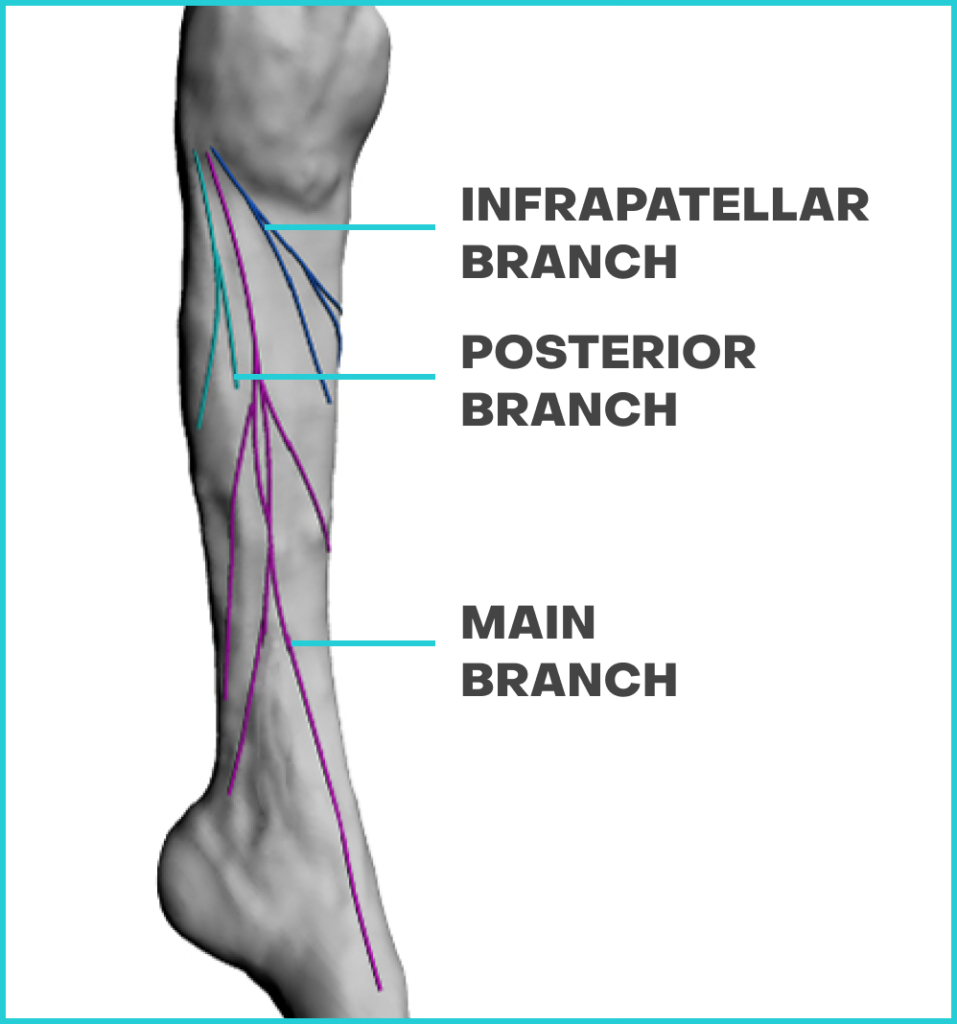
Shallow Subcutaneous / Suprafascial Location
Allows for targeted noninvasive nerve stimulation
Upper Calf Location
Easy to reach for patient
Lower Bladder Inhibition Level
83%2 lower bladder inhibition threshold vs. other targets3 allowing for less energy to deliver therapy
Afferent (Sensory) Only Nerve
More comfortable treatment experience and not limited by motor nerve side-effects
nSAFN has been studied in multiple feasibility studies in the US and Canada for OAB and other indications, with results3 equally effective or better than published results for existing invasive therapies and medications.
OAB sufferers, who number roughly 550M worldwide, have the opportunity to experience treatment without the significant side-effects that come with drugs, or the invasiveness and side-effects that come with botulinum toxin or traditional approaches to neuromodulation.

Data from three feasibility studies3 points to OAB symptom responder rates that are better than third-line therapies, with no surgery, and only mild transient side-effects
19,610,442. 9,884,187
2Moazzam, Z. & Yoo, P. (2018). Frequency-dependent inhibition of bladder function by saphenous nerve stimulation in anesthetized rats. Neurourology and urodynamics, 37(2), 592–599.
3Feasibility clinical outcomes, data on file.

© 2021 EBT MEDICAL
* Disclaimer: NiNA is currently in development and has not received market authorization for sale in the US, Canada, or any other geography.
0